Which of the Following Photosynthetic Pigments Can Be Found at the Photosystem Reaction Center?
Identify the basic components and steps of photosynthesis
The processes in all organisms—from bacteria to humans—require energy. To get this free energy, many organisms access stored energy by eating, that is, by ingesting other organisms. But where does the stored energy in nutrient originate? All of this energy can be traced dorsum to photosynthesis.
Photosynthesis is essential to all life on world; both plants and animals depend on it. Information technology is the simply biological process that can capture free energy that originates in outer infinite (sunlight) and convert it into chemic compounds (carbohydrates) that every organism uses to power its metabolism. In cursory, the energy of sunlight is captured and used to energize electrons, which are and so stored in the covalent bonds of carbohydrate molecules. How long lasting and stable are those covalent bonds? The energy extracted today by the burning of coal and petroleum products represents sunlight free energy captured and stored past photosynthesis around 300 one thousand thousand years ago.

Figure 1. Photoautotrophs including (a) plants, (b) algae, and (c) cyanobacteria synthesize their organic compounds via photosynthesis using sunlight as an energy source. Cyanobacteria and planktonic algae tin abound over enormous areas in h2o, at times completely roofing the surface. In a (d) deep bounding main vent, chemoautotrophs, such equally these (eastward) thermophilic bacteria, capture energy from inorganic compounds to produce organic compounds. The ecosystem surrounding the vents has a diverse array of animals, such every bit tubeworms, crustaceans, and octopi that derive energy from the bacteria. (credit a: modification of piece of work by Steve Hillebrand, U.S. Fish and Wild animals Service; credit b: modification of work past "eutrophication&hypoxia"/Flickr; credit c: modification of work past NASA; credit d: University of Washington, NOAA; credit e: modification of work by Mark Amend, Due west Coast and Polar Regions Undersea Research Center, UAF, NOAA)

Effigy 2. The energy stored in saccharide molecules from photosynthesis passes through the food concatenation. The predator that eats these deer receives a portion of the free energy that originated in the photosynthetic vegetation that the deer consumed. (credit: modification of work past Steve VanRiper, U.S. Fish and Wildlife Service)
Plants, algae, and a group of bacteria called cyanobacteria are the only organisms capable of performing photosynthesis (Figure 1). Considering they utilise light to manufacture their own food, they are chosen photoautotrophs (literally, "self-feeders using light"). Other organisms, such as animals, fungi, and virtually other leaner, are termed heterotrophs ("other feeders"), because they must rely on the sugars produced by photosynthetic organisms for their energy needs. A third very interesting group of bacteria synthesize sugars, not past using sunlight'southward free energy, but by extracting energy from inorganic chemical compounds; hence, they are referred to as chemoautotrophs.
The importance of photosynthesis is non just that it can capture sunlight'due south free energy. A lizard sunning itself on a cold 24-hour interval tin can apply the sun's free energy to warm up. Photosynthesis is vital because it evolved equally a way to shop the energy in solar radiations (the "photo" part) as high-free energy electrons in the carbon-carbon bonds of saccharide molecules (the "synthesis" function). Those carbohydrates are the energy source that heterotrophs use to power the synthesis of ATP via respiration. Therefore, photosynthesis powers 99 percent of Earth'south ecosystems. When a top predator, such every bit a wolf, preys on a deer (Figure two), the wolf is at the cease of an energy path that went from nuclear reactions on the surface of the lord's day, to light, to photosynthesis, to vegetation, to deer, and finally to wolf.
Learning Objectives
- Identify the reactants and products of photosynthesis
- Depict the visible and electromagnetic spectrums of light as they applies to photosynthesis
- Describe the low-cal-dependent reactions that accept place during photosynthesis
- Place the light-independent reactions in photosynthesis
Photosynthesis is a multi-stride process that requires sunlight, carbon dioxide (which is low in energy), and water as substrates (Figure 3). After the procedure is complete, information technology releases oxygen and produces glyceraldehyde-3-phosphate (GA3P), simple sugar molecules (which are high in free energy) that can subsequently exist converted into glucose, sucrose, or any of dozens of other carbohydrate molecules. These sugar molecules comprise energy and the energized carbon that all living things demand to survive.

Figure 3. Photosynthesis uses solar energy, carbon dioxide, and water to produce energy-storing carbohydrates. Oxygen is generated as a waste product of photosynthesis.
The post-obit is the chemical equation for photosynthesis (Figure iv):
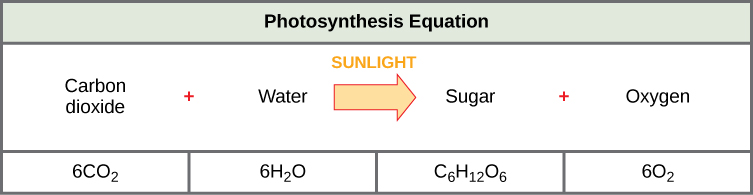
Figure four. The basic equation for photosynthesis is deceptively uncomplicated. In reality, the procedure takes place in many steps involving intermediate reactants and products. Glucose, the principal free energy source in cells, is made from ii 3-carbon GA3Ps.
Although the equation looks simple, the many steps that take place during photosynthesis are actually quite complex. Before learning the details of how photoautotrophs plow sunlight into food, it is important to become familiar with the structures involved.
In plants, photosynthesis generally takes identify in leaves, which consist of several layers of cells. The process of photosynthesis occurs in a middle layer called themesophyll. The gas substitution of carbon dioxide and oxygen occurs through small, regulated openings called stomata (singular: stoma), which likewise play roles in the regulation of gas exchange and water rest. The stomata are typically located on the underside of the foliage, which helps to minimize h2o loss. Each stoma is flanked past guard cells that regulate the opening and endmost of the stomata by swelling or shrinking in response to osmotic changes.
In all autotrophic eukaryotes, photosynthesis takes place inside an organelle called achloroplast. For plants, chloroplast-containing cells exist in the mesophyll. Chloroplasts have a double membrane envelope (composed of an outer membrane and an inner membrane). Within the chloroplast are stacked, disc-shaped structures called thylakoids. Embedded in the thylakoid membrane is chlorophyll, a pigment (molecule that absorbs low-cal) responsible for the initial interaction between light and constitute material, and numerous proteins that brand up the electron transport concatenation. The thylakoid membrane encloses an internal space called the thylakoid lumen. Every bit shown in Figure 5, a stack of thylakoids is chosen a granum, and the liquid-filled space surrounding the granum is called stroma or "bed" (not to be confused with stoma or "rima oris," an opening on the leaf epidermis).
Practice Question
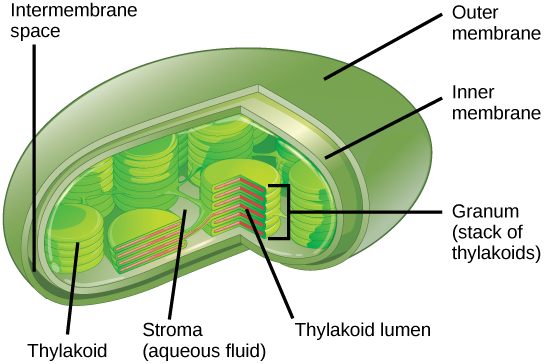
Effigy 5. Photosynthesis takes identify in chloroplasts, which take an outer membrane and an inner membrane. Stacks of thylakoids called grana form a tertiary membrane layer.
On a hot, dry day, plants close their stomata to conserve water. What affect will this have on photosynthesis?
Bear witness Answer
Levels of carbon dioxide (a necessary photosynthetic substrate) will immediately autumn. As a result, the rate of photosynthesis volition exist inhibited.
The 2 Parts of Photosynthesis
Photosynthesis takes place in 2 sequential stages: the light-dependent reactions and the light independent-reactions. In thelight-dependent reactions, free energy from sunlight is absorbed past chlorophyll and that energy is converted into stored chemical energy. In the calorie-free-independent reactions, the chemical energy harvested during the light-dependent reactions drive the assembly of sugar molecules from carbon dioxide. Therefore, although the light-independent reactions practise not use low-cal every bit a reactant, they crave the products of the low-cal-dependent reactions to function. In addition, several enzymes of the low-cal-independent reactions are activated by light. The light-dependent reactions utilize sure molecules to temporarily store the energy: These are referred to as energy carriers. The energy carriers that motion energy from light-dependent reactions to light-independent reactions tin exist thought of as "full" because they are rich in free energy. Later on the energy is released, the "empty" energy carriers return to the lite-dependent reaction to obtain more energy. Figure 6 illustrates the components inside the chloroplast where the light-dependent and light-contained reactions take place.
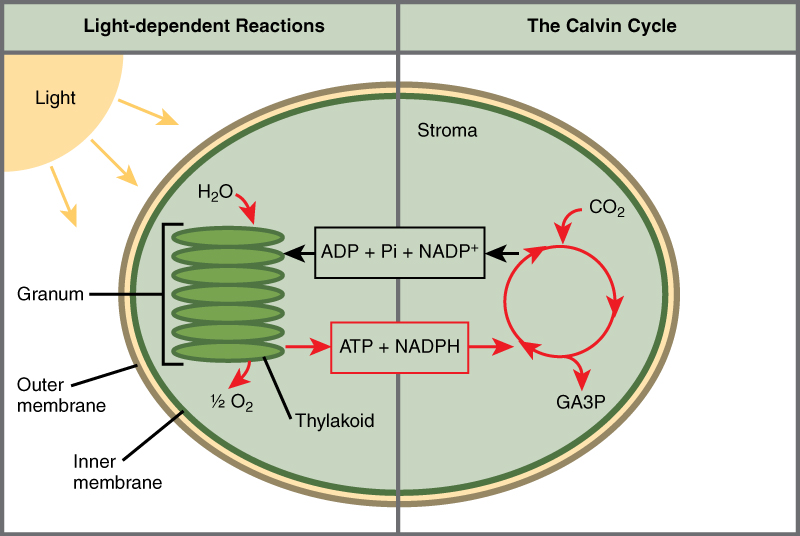
Figure half dozen. Photosynthesis takes place in 2 stages: low-cal dependent reactions and the Calvin cycle. Light-dependent reactions, which take place in the thylakoid membrane, use calorie-free free energy to make ATP and NADPH. The Calvin cycle, which takes identify in the stroma, uses energy derived from these compounds to make GA3P from CO2.
Photosynthesis at the Grocery Store

Effigy 7. Foods that humans consume originate from photosynthesis. (credit: Associação Brasileira de Supermercados)
Major grocery stores in the United States are organized into departments, such as dairy, meats, produce, bread, cereals, and then forth. Each alley (Figure 7) contains hundreds, if not thousands, of unlike products for customers to purchase and consume.
Although there is a large variety, each item links back to photosynthesis. Meats and dairy link because the animals were fed institute-based foods. The breads, cereals, and pastas come largely from starchy grains, which are the seeds of photosynthesis-dependent plants. What about desserts and drinks? All of these products contain sugar—sucrose is a found product, a disaccharide, a carbohydrate molecule, which is built directly from photosynthesis. Moreover, many items are less plainly derived from plants: for example, paper appurtenances are generally plant products, and many plastics (arable as products and packaging) can be derived from algae or from oil, the fossilized remains of photosynthetic organisms. Virtually every spice and flavoring in the spice aisle was produced by a plant as a leafage, root, bark, bloom, fruit, or stem. Ultimately, photosynthesis connects to every meal and every nutrient a person consumes.
Spectrums of Light
How tin can lite be used to make food? When a person turns on a lamp, electrical energy becomes light energy. Like all other forms of kinetic free energy, light tin travel, alter form, and be harnessed to do work. In the case of photosynthesis, light energy is converted into chemic energy, which photoautotrophs use to build carbohydrate molecules. Nonetheless, autotrophs only utilize a few specific components of sunlight.
What Is Light Energy?
The sun emits an enormous amount of electromagnetic radiation (solar energy). Humans can encounter only a fraction of this energy, which portion is therefore referred to as "visible light." The manner in which solar energy travels is described as waves. Scientists can make up one's mind the amount of energy of a wave by measuring its wavelength, the distance betwixt sequent points of a wave. A unmarried wave is measured from two consecutive points, such equally from crest to crest or from trough to trough (Figure 8).
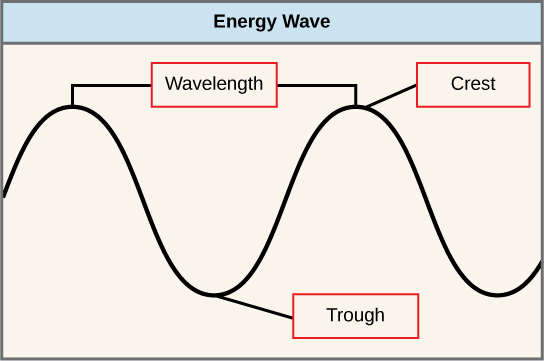
Figure eight. The wavelength of a unmarried wave is the altitude between two consecutive points of similar position (two crests or two troughs) along the moving ridge.
Visible light constitutes only 1 of many types of electromagnetic radiation emitted from the sun and other stars. Scientists differentiate the diverse types of radiant energy from the dominicus within the electromagnetic spectrum. The electromagnetic spectrum is the range of all possible frequencies of radiation (Figure 9). The difference between wavelengths relates to the corporeality of energy carried by them.
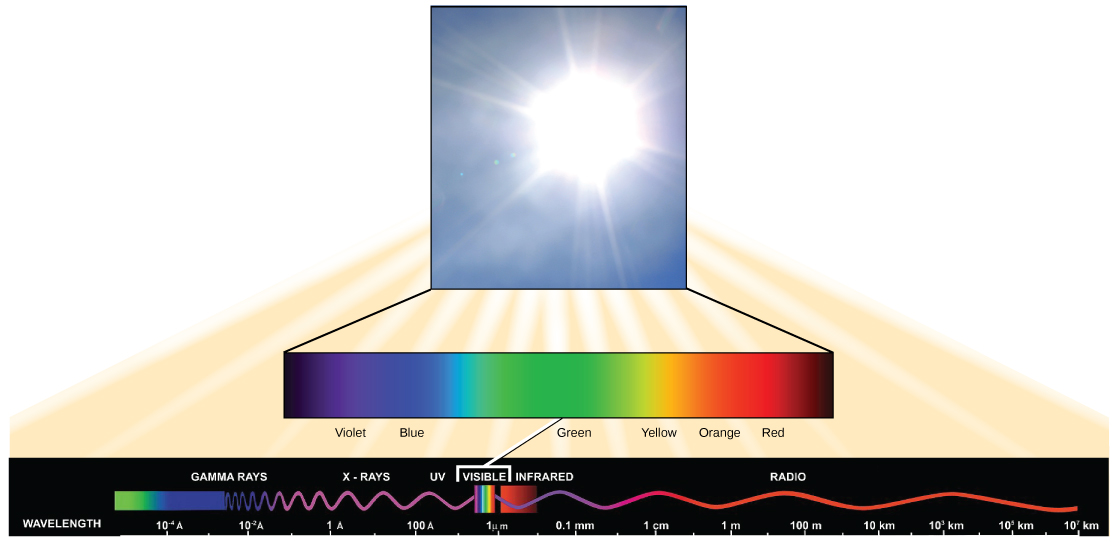
Effigy nine. The dominicus emits free energy in the course of electromagnetic radiation. This radiation exists at different wavelengths, each of which has its own characteristic energy. All electromagnetic radiation, including visible lite, is characterized by its wavelength.
Each type of electromagnetic radiation travels at a item wavelength. The longer the wavelength (or the more stretched out it appears in the diagram), the less free energy is carried. Brusk, tight waves acquit the most energy. This may seem illogical, only think of information technology in terms of a piece of moving a heavy rope. Information technology takes little effort by a person to move a rope in long, broad waves. To make a rope motility in short, tight waves, a person would demand to utilise significantly more than free energy.
The electromagnetic spectrum (Figure 9) shows several types of electromagnetic radiation originating from the lord's day, including Ten-rays and ultraviolet (UV) rays. The college-energy waves tin can penetrate tissues and harm cells and Dna, explaining why both 10-rays and UV rays can be harmful to living organisms.
Absorption of Light
Lite energy initiates the process of photosynthesis when pigments blot the light. Organic pigments, whether in the human retina or the chloroplast thylakoid, have a narrow range of energy levels that they can absorb. Free energy levels lower than those represented by red lite are bereft to raise an orbital electron to a populatable, excited (quantum) state. Energy levels college than those in bluish light volition physically tear the molecules autonomously, called bleaching. Then retinal pigments can only "see" (blot) 700 nm to 400 nm light, which is therefore called visible light. For the same reasons, plants pigment molecules absorb just light in the wavelength range of 700 nm to 400 nm; plant physiologists refer to this range for plants as photosynthetically active radiation.
The visible calorie-free seen by humans equally white low-cal really exists in a rainbow of colors. Certain objects, such equally a prism or a drop of water, disperse white light to reveal the colors to the human being eye. The visible light portion of the electromagnetic spectrum shows the rainbow of colors, with violet and blue having shorter wavelengths, and therefore higher energy. At the other end of the spectrum toward cherry, the wavelengths are longer and accept lower energy (Figure 10).
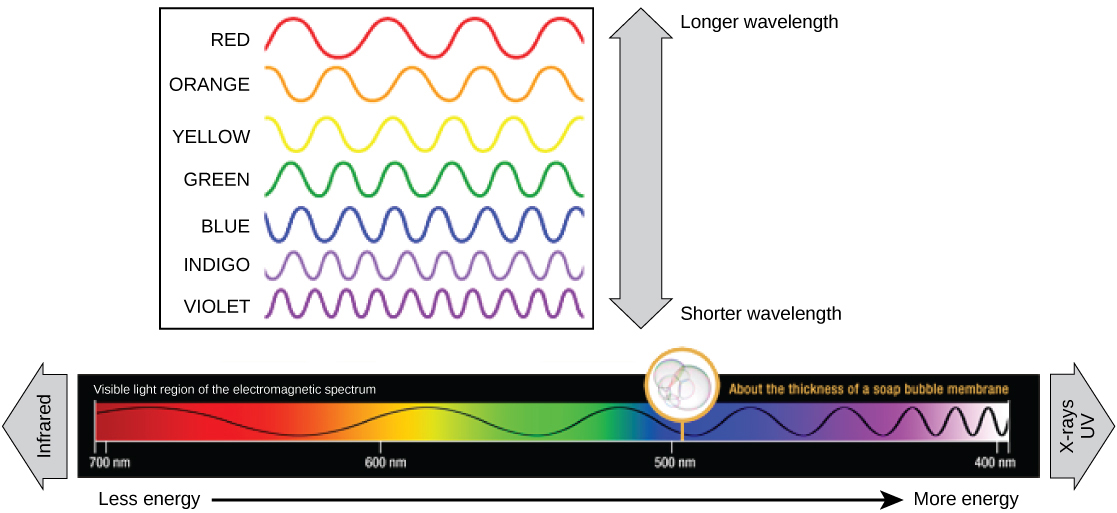
Figure x. The colors of visible light do not behave the same amount of energy. Violet has the shortest wavelength and therefore carries the most free energy, whereas ruddy has the longest wavelength and carries the least amount of energy. (credit: modification of work past NASA)
Understanding Pigments
Dissimilar kinds of pigments exist, and each has evolved to blot simply certain wavelengths (colors) of visible low-cal. Pigments reflect or transmit the wavelengths they cannot blot, making them announced in the respective color.
Chlorophylls and carotenoids are the 2 major classes of photosynthetic pigments found in plants and algae; each class has multiple types of pigment molecules. At that place are five major chlorophylls:a, b, c and d and a related molecule found in prokaryotes called bacteriochlorophyll. Chlorophyll a and chlorophyll b are establish in college plant chloroplasts and will be the focus of the following discussion.
With dozens of dissimilar forms, carotenoids are a much larger group of pigments. The carotenoids establish in fruit—such every bit the red of lycopersicon esculentum (lycopene), the yellowish of corn seeds (zeaxanthin), or the orange of an orangish skin (β-carotene)—are used as advertisements to attract seed dispersers. In photosynthesis,
carotenoids office as photosynthetic pigments that are very efficient molecules for the disposal of excess free energy. When a leaf is exposed to full sun, the light-dependent reactions are required to process an enormous corporeality of energy; if that free energy is non handled properly, it can do significant damage. Therefore, many carotenoids reside in the thylakoid membrane, absorb excess energy, and safely dissipate that free energy as estrus.
Each type of paint can be identified past the specific pattern of wavelengths it absorbs from visible light, which is theabsorption spectrum. The graph in Effigy xi shows the absorption spectra for chlorophylla, chlorophyll b, and a type of carotenoid pigment called β-carotene (which absorbs blue and green light). Detect how each pigment has a singled-out set of peaks and troughs, revealing a highly specific design of absorption. Chlorophyll a absorbs wavelengths from either end of the visible spectrum (blue and scarlet), but not green. Because light-green is reflected or transmitted, chlorophyll appears green. Carotenoids absorb in the short-wavelength bluish region, and reflect the longer xanthous, red, and orange wavelengths.
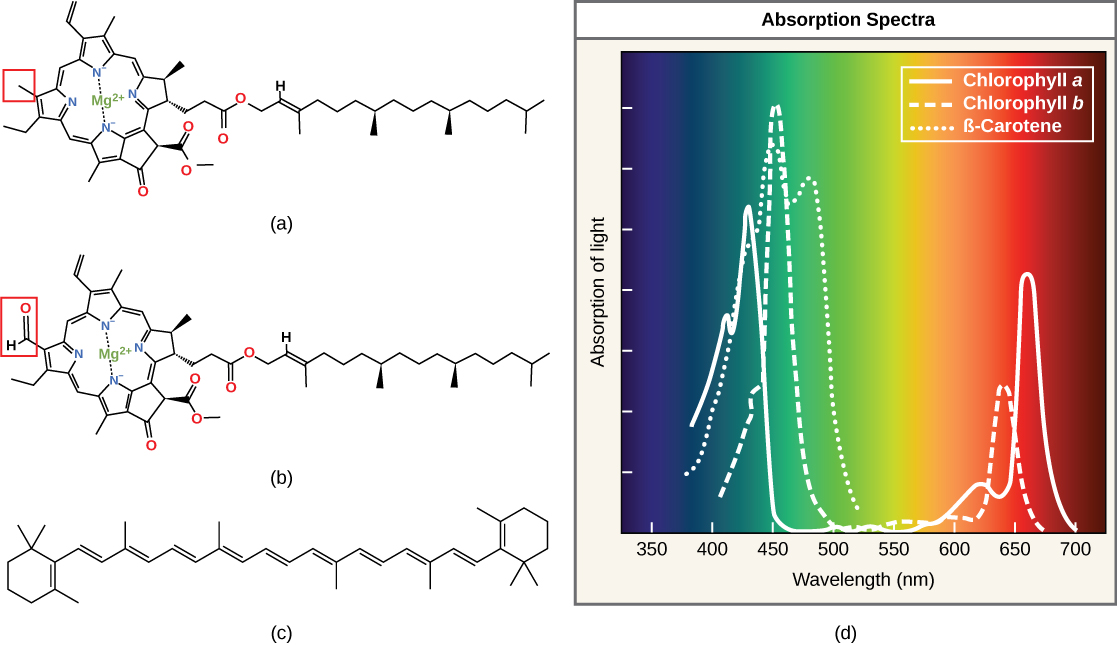
Effigy eleven. (a) Chlorophyll a, (b) chlorophyll b, and (c) β-carotene are hydrophobic organic pigments plant in the thylakoid membrane. Chlorophyll a and b, which are identical except for the role indicated in the red box, are responsible for the dark-green color of leaves. β-carotene is responsible for the orange color in carrots. Each pigment has (d) a unique absorbance spectrum.

Figure 12. Plants that commonly grow in the shade have adjusted to low levels of light by changing the relative concentrations of their chlorophyll pigments. (credit: Jason Hollinger)
Many photosynthetic organisms have a mixture of pigments; using them, the organism tin absorb energy from a wider range of wavelengths. Not all photosynthetic organisms accept full admission to sunlight. Some organisms grow underwater where lite intensity and quality decrease and change with depth. Other organisms abound in contest for light. Plants on the rainforest floor must be able to absorb whatever fleck of light that comes through, because the taller copse absorb near of the sunlight and scatter the remaining solar radiation (Figure 12).
When studying a photosynthetic organism, scientists tin determine the types of pigments nowadays by generating absorption spectra. An musical instrument called aspectrophotometer tin differentiate which wavelengths of lite a substance can blot. Spectrophotometers mensurate transmitted light and compute from it the assimilation. By extracting pigments from leaves and placing these samples into a spectrophotometer, scientists can identify which wavelengths of light an organism can absorb. Additional methods for the identification of plant pigments include diverse types of chromatography that separate the pigments by their relative affinities to solid and mobile phases.
Light-Dependent Reactions
The overall function of light-dependent reactions is to convert solar free energy into chemical energy in the form of NADPH and ATP. This chemical energy supports the light-independent reactions and fuels the assembly of sugar molecules. The lite-dependent reactions are depicted in Figure xiii. Protein complexes and paint molecules work together to produce NADPH and ATP.
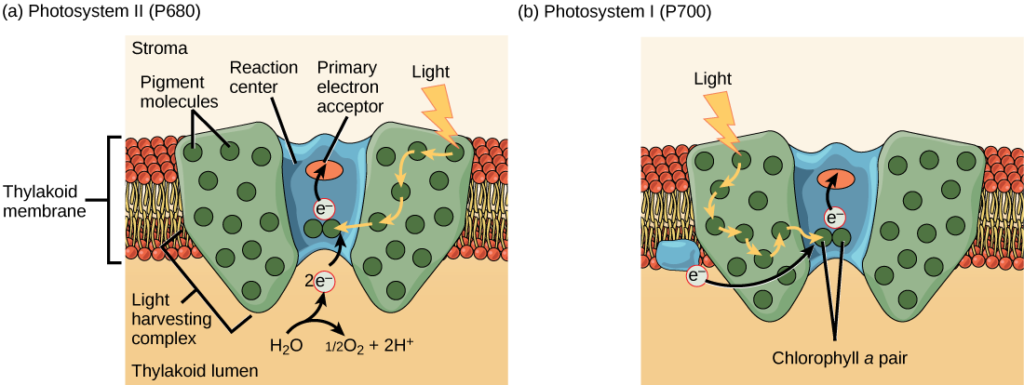
Effigy 13. A photosystem consists of a light-harvesting complex and a reaction center. Pigments in the light-harvesting complex laissez passer light energy to two special chlorophyll a molecules in the reaction middle. The light excites an electron from the chlorophyll a pair, which passes to the chief electron acceptor. The excited electron must then be replaced. In (a) photosystem Ii, the electron comes from the splitting of water, which releases oxygen as a waste product. In (b) photosystem I, the electron comes from the chloroplast electron transport concatenation discussed below.
The actual stride that converts calorie-free energy into chemic energy takes place in a multiprotein complex called aphotosystem, two types of which are establish embedded in the thylakoid membrane, photosystem Ii (PSII) and photosystem I (PSI) (Figure xiv). The ii complexes differ on the basis of what they oxidize (that is, the source of the low-energy electron supply) and what they reduce (the identify to which they deliver their energized electrons).
Both photosystems have the same bones construction; a number of antenna proteins to which the chlorophyll molecules are bound surround the reaction center where the photochemistry takes place. Each photosystem is serviced by the light-harvesting circuitous, which passes energy from sunlight to the reaction centre; information technology consists of multiple antenna proteins that contain a mixture of 300–400 chlorophylla and b molecules likewise as other pigments like carotenoids. The absorption of a single photon or distinct quantity or "packet" of low-cal by any of the chlorophylls pushes that molecule into an excited state. In short, the calorie-free energy has now been captured by biological molecules but is not stored in any useful form yet. The energy is transferred from chlorophyll to chlorophyll until eventually (after about a millionth of a 2d), it is delivered to the reaction center. Up to this point, only energy has been transferred between molecules, not electrons.
Practise Question
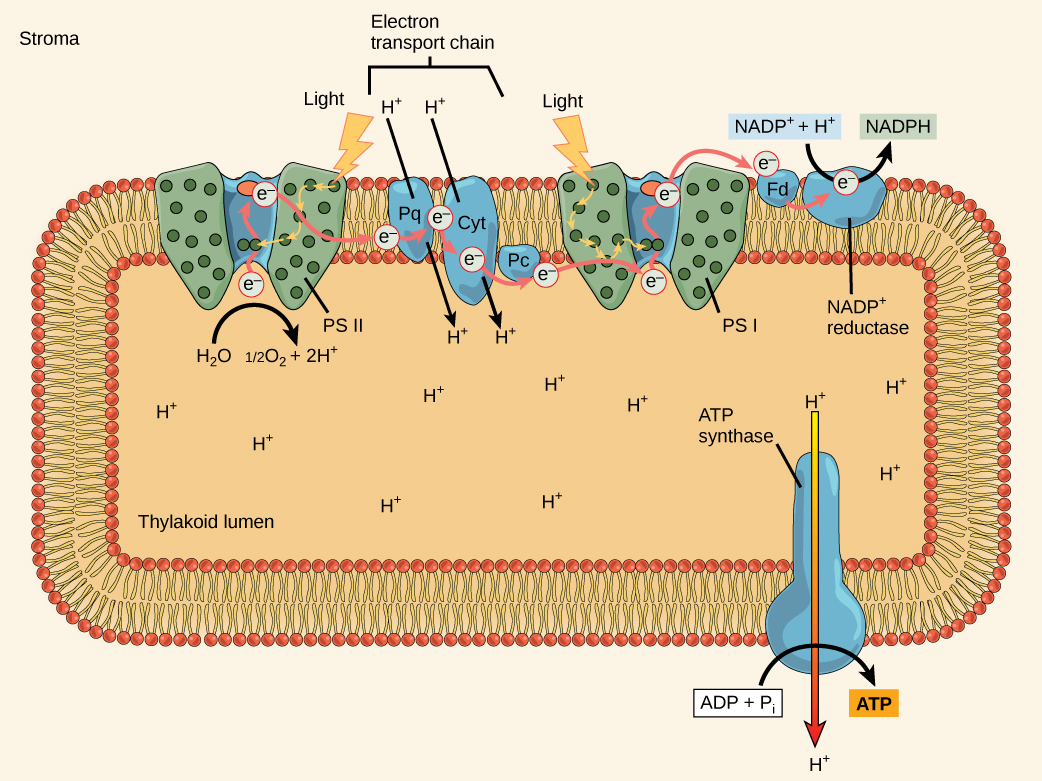
Figure 14. The photosystem II (PSII) reaction eye and the photosystem I (PSI).
In the photosystem II (PSII) reaction center, free energy from sunlight is used to extract electrons from water. The electrons travel through the chloroplast electron send chain to photosystem I (PSI), which reduces NADP+ to NADPH. The electron transport chain moves protons across the thylakoid membrane into the lumen. At the same time, splitting of water adds protons to the lumen, and reduction of NADPH removes protons from the stroma. The internet result is a low pH in the thylakoid lumen, and a high pH in the stroma. ATP synthase uses this electrochemical gradient to make ATP. What is the initial source of electrons for the chloroplast electron transport chain?
- water
- oxygen
- carbon dioxide
- NADPH
Show Answer
Water is the initial source.
The reaction center contains a pair of chlorophylla molecules with a special property. Those two chlorophylls can undergo oxidation upon excitation; they can really surrender an electron in a procedure chosen a photoact. It is at this footstep in the reaction center, that low-cal energy is converted into an excited electron. All of the subsequent steps involve getting that electron onto the free energy carrier NADPH for delivery to the Calvin cycle where the electron is deposited onto carbon for long-term storage in the form of a carbohydrate. PSII and PSI are two major components of the photosynthetic electron send concatenation, which also includes the cytochrome complex. The cytochrome complex, an enzyme composed of two protein complexes, transfers the electrons from the carrier molecule plastoquinone (Pq) to the protein plastocyanin (Pc), thus enabling both the transfer of protons across the thylakoid membrane and the transfer of electrons from PSII to PSI.
The reaction heart of PSII (calledP680) delivers its high-energy electrons, 1 at the time, to the primary electron acceptor, and through the electron send concatenation (Pq to cytochrome complex to plastocyanine) to PSI. P680's missing electron is replaced by extracting a depression-energy electron from water; thus, water is split and PSII is re-reduced after every photoact. Splitting i H2O molecule releases two electrons, two hydrogen atoms, and ane atom of oxygen. Splitting two molecules is required to course 1 molecule of diatomic O2 gas. About 10 percent of the oxygen is used by mitochondria in the leaf to support oxidative phosphorylation. The remainder escapes to the temper where it is used past aerobic organisms to support respiration.
As electrons move through the proteins that reside between PSII and PSI, they lose free energy. That energy is used to motility hydrogen atoms from the stromal side of the membrane to the thylakoid lumen. Those hydrogen atoms, plus the ones produced by splitting h2o, accumulate in the thylakoid lumen and will be used synthesize ATP in a later footstep. Because the electrons have lost free energy prior to their inflow at PSI, they must be re-energized by PSI, hence, some other photon is absorbed past the PSI antenna. That energy is relayed to the PSI reaction center (calledP700). P700 is oxidized and sends a loftier-energy electron to NADP+ to form NADPH. Thus, PSII captures the free energy to create proton gradients to make ATP, and PSI captures the energy to reduce NADP+ into NADPH. The two photosystems work in concert, in role, to guarantee that the product of NADPH volition roughly equal the production of ATP. Other mechanisms exist to fine melody that ratio to exactly friction match the chloroplast's constantly changing energy needs.
Generating an Energy Carrier: ATP
As in the intermembrane infinite of the mitochondria during cellular respiration, the buildup of hydrogen ions inside the thylakoid lumen creates a concentration gradient. The passive diffusion of hydrogen ions from high concentration (in the thylakoid lumen) to low concentration (in the stroma) is harnessed to create ATP, only every bit in the electron transport chain of cellular respiration. The ions build up energy because of diffusion and because they all have the aforementioned electrical charge, repelling each other.
To release this energy, hydrogen ions will rush through any opening, like to water jetting through a hole in a dam. In the thylakoid, that opening is a passage through a specialized protein channel called the ATP synthase. The energy released by the hydrogen ion stream allows ATP synthase to adhere a third phosphate group to ADP, which forms a molecule of ATP (Effigy xiv). The menstruation of hydrogen ions through ATP synthase is chosen chemiosmosis considering the ions move from an area of loftier to an area of depression concentration through a semi-permeable structure.
Calorie-free-Contained Reactions
After the energy from the lord's day is converted into chemical energy and temporarily stored in ATP and NADPH molecules, the prison cell has the fuel needed to build sugar molecules for long-term energy storage. The products of the low-cal-dependent reactions, ATP and NADPH, have lifespans in the range of millionths of seconds, whereas the products of the light-independent reactions (carbohydrates and other forms of reduced carbon) can survive for hundreds of millions of years. The carbohydrate molecules made will have a backbone of carbon atoms. Where does the carbon come from? It comes from carbon dioxide, the gas that is a waste product of respiration in microbes, fungi, plants, and animals.
In plants, carbon dioxide (CO2) enters the leaves through stomata, where information technology diffuses over short distances through intercellular spaces until information technology reaches the mesophyll cells. One time in the mesophyll cells, CO2 diffuses into the stroma of the chloroplast—the site of lite-independent reactions of photosynthesis. These reactions actually have several names associated with them. Another term, the Calvin bike, is named for the man who discovered information technology, and considering these reactions function as a cycle. Others call information technology the Calvin-Benson cycle to include the name of another scientist involved in its discovery. The about outdated name is night reactions, because light is not directly required (Figure fifteen). However, the term dark reaction tin be misleading because it implies incorrectly that the reaction simply occurs at night or is independent of light, which is why most scientists and instructors no longer apply information technology.
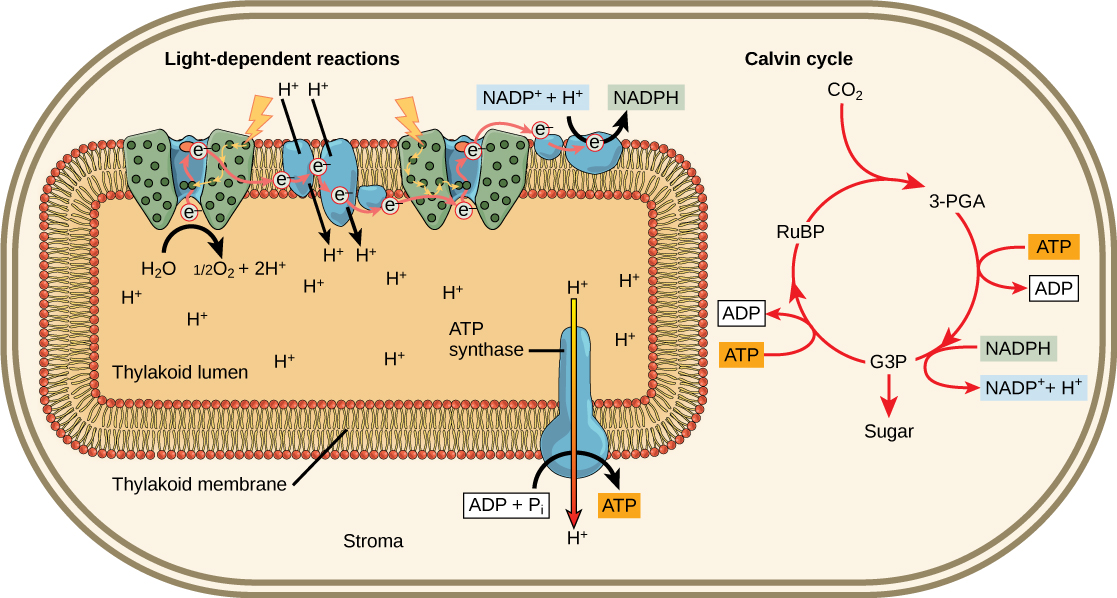
Figure fifteen. Low-cal reactions harness free energy from the sun to produce chemic bonds, ATP, and NADPH. These free energy-carrying molecules are made in the stroma where carbon fixation takes place.
The lite-independent reactions of the Calvin cycle can be organized into 3 basic stages: fixation, reduction, and regeneration.
Phase i: Fixation
In the stroma, in improver to COii, two other components are present to initiate the lite-independent reactions: an enzyme called ribulose bisphosphate carboxylase (RuBisCO), and three molecules of ribulose bisphosphate (RuBP), as shown in Figure 16. RuBP has v atoms of carbon, flanked by two phosphates.
Practice Question
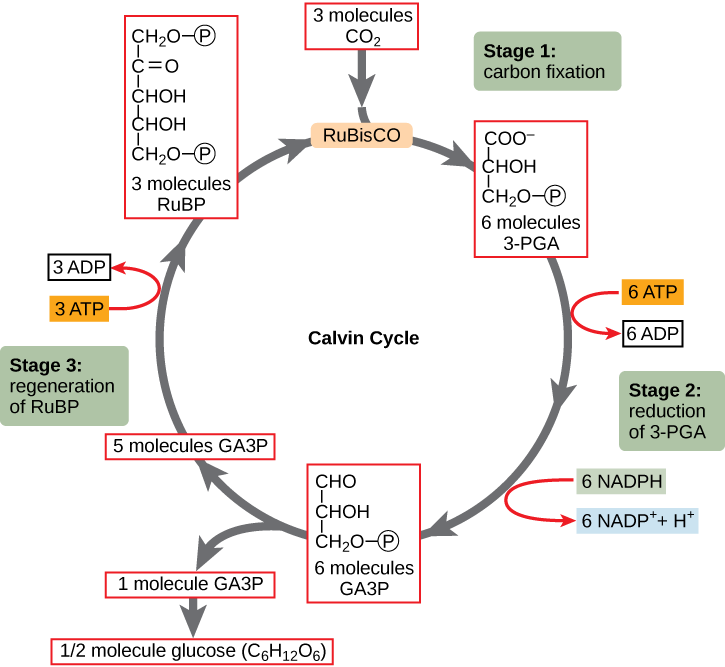
Figure 16. The Calvin bike has iii stages.
In phase 1, the enzyme RuBisCO incorporates carbon dioxide into an organic molecule, 3-PGA. In stage two, the organic molecule is reduced using electrons supplied by NADPH. In stage 3, RuBP, the molecule that starts the cycle, is regenerated so that the bicycle can continue. Only 1 carbon dioxide molecule is incorporated at a time, so the cycle must be completed 3 times to produce a single three-carbon GA3P molecule, and six times to produce a vi-carbon glucose molecule.
Which of the following statements is true?
- In photosynthesis, oxygen, carbon dioxide, ATP, and NADPH are reactants. GA3P and water are products.
- In photosynthesis, chlorophyll, water, and carbon dioxide are reactants. GA3P and oxygen are products.
- In photosynthesis, water, carbon dioxide, ATP, and NADPH are reactants. RuBP and oxygen are products.
- In photosynthesis, water and carbon dioxide are reactants. GA3P and oxygen are products.
Evidence Respond
Answer d is truthful.
RuBisCO catalyzes a reaction between COii and RuBP. For each COtwo molecule that reacts with 1 RuBP, two molecules of another compound (three-PGA) form. PGA has three carbons and one phosphate. Each plow of the bicycle involves only one RuBP and 1 carbon dioxide and forms two molecules of 3-PGA. The number of carbon atoms remains the aforementioned, equally the atoms motility to form new bonds during the reactions (3 atoms from 3CO2 + 15 atoms from 3RuBP = eighteen atoms in iii atoms of 3-PGA). This procedure is calledcarbon fixation, because COtwo is "fixed" from an inorganic form into organic molecules.
Stage 2: Reduction
ATP and NADPH are used to convert the 6 molecules of 3-PGA into vi molecules of a chemic called glyceraldehyde 3-phosphate (G3P). That is a reduction reaction considering information technology involves the proceeds of electrons by iii-PGA. Think that areduction is the gain of an electron by an cantlet or molecule. Six molecules of both ATP and NADPH are used. For ATP, energy is released with the loss of the terminal phosphate atom, converting it into ADP; for NADPH, both energy and a hydrogen atom are lost, converting information technology into NADP+. Both of these molecules return to the nearby light-dependent reactions to be reused and reenergized.
Stage 3: Regeneration
Interestingly, at this indicate, merely one of the G3P molecules leaves the Calvin cycle and is sent to the cytoplasm to contribute to the formation of other compounds needed by the establish. Considering the G3P exported from the chloroplast has three carbon atoms, it takes three "turns" of the Calvin cycle to fix enough net carbon to export i G3P. But each plow makes two G3Ps, thus three turns brand six G3Ps. Ane is exported while the remaining v G3P molecules remain in the cycle and are used to regenerate RuBP, which enables the system to prepare for more COii to be fixed. Iii more molecules of ATP are used in these regeneration reactions.
This link leads to an animation of the Calvin bike. Click stage 1, stage 2, and then stage 3 to see G3P and ATP regenerate to grade RuBP.
Development of Photosynthesis

Effigy 17. The harsh conditions of the desert have led plants like these cacti to evolve variations of the light-independent reactions of photosynthesis. These variations increase the efficiency of water usage, helping to conserve h2o and energy. (credit: Piotr Wojtkowski)
During the evolution of photosynthesis, a major shift occurred from the bacterial type of photosynthesis that involves only ane photosystem and is typically anoxygenic (does non generate oxygen) into modern oxygenic (does generate oxygen) photosynthesis, employing 2 photosystems. This mod oxygenic photosynthesis is used by many organisms—from behemothic tropical leaves in the rainforest to tiny cyanobacterial cells—and the process and components of this photosynthesis remain largely the same. Photosystems absorb calorie-free and use electron transport bondage to catechumen free energy into the chemical free energy of ATP and NADH. The subsequent light-independent reactions then assemble carbohydrate molecules with this energy.
Photosynthesis in desert plants has evolved adaptations that conserve water. In the harsh dry out heat, every drop of water must be used to survive. Because stomata must open to permit for the uptake of CO2, water escapes from the foliage during active photosynthesis. Desert plants have evolved processes to conserve water and deal with harsh conditions. A more than efficient use of CO2 allows plants to arrange to living with less h2o. Some plants such every bit cacti (Figure 17) tin can prepare materials for photosynthesis during the dark by a temporary carbon fixation/storage procedure, because opening the stomata at this time conserves water due to cooler temperatures. In improver, cacti take evolved the ability to carry out low levels of photosynthesis without opening stomata at all, a mechanism to face extremely dry periods.
Now that we've learned nigh the different pieces of photosynthesis, allow'southward put information technology all together. This video walks y'all through the process of photosynthesis as a whole:
In Summary: An Overview of Photosynthesis
The process of photosynthesis transformed life on World. By harnessing energy from the sunday, photosynthesis evolved to let living things admission to enormous amounts of energy. Because of photosynthesis, living things gained access to sufficient energy that allowed them to build new structures and attain the biodiversity evident today.
But certain organisms, chosen photoautotrophs, tin perform photosynthesis; they require the presence of chlorophyll, a specialized pigment that absorbs sure portions of the visible spectrum and can capture energy from sunlight. Photosynthesis uses carbon dioxide and water to assemble carbohydrate molecules and release oxygen as a waste product into the temper. Eukaryotic autotrophs, such every bit plants and algae, accept organelles chosen chloroplasts in which photosynthesis takes place, and starch accumulates. In prokaryotes, such as blue-green alga, the process is less localized and occurs within folded membranes, extensions of the plasma membrane, and in the cytoplasm.
The pigments of the first part of photosynthesis, the light-dependent reactions, absorb free energy from sunlight. A photon strikes the antenna pigments of photosystem Ii to initiate photosynthesis. The energy travels to the reaction eye that contains chlorophylla to the electron ship chain, which pumps hydrogen ions into the thylakoid interior. This activity builds upwards a high concentration of ions. The ions catamenia through ATP synthase via chemiosmosis to class molecules of ATP, which are used for the formation of sugar molecules in the second stage of photosynthesis. Photosystem I absorbs a second photon, which results in the formation of an NADPH molecule, some other energy and reducing power carrier for the light-independent reactions.
Cheque Your Understanding
Answer the question(s) below to see how well yous understand the topics covered in the previous section. This short quiz doesnot count toward your course in the form, and you can retake information technology an unlimited number of times.
Employ this quiz to check your understanding and decide whether to (1) study the previous section further or (2) movement on to the next department.
Source: https://courses.lumenlearning.com/suny-wmopen-biology1/chapter/photosynthesis/
0 Response to "Which of the Following Photosynthetic Pigments Can Be Found at the Photosystem Reaction Center?"
Post a Comment